Why have many molluscs lost or reduced their shells?
The dramatic 'explosion' of hard-shelled animals into the fossil record that occurred 500 million years ago clearly suggests that a tough shell is a useful accessory for an animal to have. It provides a reliable means of protection, both against predators and against the physical environment, and it can be modified for many other purposes (some creatures, for instance, have converted their shells into gas-filled flotation aids). The phylum that has undoubtedly been most successful in exploiting the advantages of having a hard, calcified covering is the molluscs, yet there are several types of mollusc whose shells have been greatly reduced in size, or even lost completely, during their evolution. In this essay, I will examine two major groups of molluscs within which secondarily shell-less forms have evolved, in an attempt to discover the reasons behind this puzzling characteristic.
The gastropods
With over 30,000 species surviving in a wide variety of habitats, the gastropods are by far the most successful class of living molluscs. Snails, with their familiar coiled shells, probably typify the ancestral gastropod body plan, but the group also includes a diverse assortment of 'slugs' (both terrestrial and marine), in which the shell has become greatly reduced, or is absent completely. In many lineages, there are groups of related species that nicely illustrate the trend towards reduction, then internalisation, and finally loss of the shell.
This reduction and loss of shells was not an isolated evolutionary event; it is seen in at least ten different orders of gastropods, most of which are believed to have evolved the trait independently. The parallel evolution of a similar characteristic in separate lineages is usually interpreted as powerful evidence that the adaptation confers a strong evolutionary advantage, so what benefit was it that prompted so many different gastropod groups to abandon their protective shells?
Gastropods are the only molluscs to have successfully colonised the land, and their ability to withdraw into protective, waterproof shells undoubtedly played a part in this achievement. However, the construction of a thick shell requires calcium, and this vital element is often scarce in terrestrial ecosystems. Land snails therefore have very thin shells by comparison with their marine relatives. The total shell loss that is seen in terrestrial slugs may be an adaptation to cope with a lack of calcium, and there is evidence that the original distribution of slugs was confined to low-calcium environments. Slugs are largely restricted to humid environments, too, since without shells they are extremely vulnerable to dehydration, in spite of water-conserving adaptations such as nocturnal behaviour and a remarkable physiological ability to tolerate desiccation. Slugs do, nonetheless, have several major advantages over snails: in the absence of shells, not only do they economise on calcium, but they can crawl into tighter spaces, burrow more easily in search of prey, and distend their bodies to swallow large meals.
There is always plenty of calcium in the sea, so the fact that many marine gastropods have also lost their shells cannot be explained by mineral deficiency. Instead, the slug-like body form of such molluscs may be an adaptation to a burrowing or swimming lifestyle, in which shell reduction is necessary for streamlining, and (in the case of pelagic forms) reducing the weight of the animal. In some sea slugs, exposing the whole body to sunlight (rather than covering it in a shell) confers a more bizarre advantage: the ability to photosynthesise! These species either contain chloroplasts (absorbed from algae eaten by the slug), or harbour entire colonies of symbiotic algae (zooxanthellae), allowing them to utilise energy directly from the sun.
Loss of the shell obviously leaves a mollusc vulnerable to attack, but sea slugs have evolved a variety of alternative means to deter predators. Some secrete toxins (or sequester toxins from their diet), some gain protection by mimicking unpalatable species, some are well-camouflaged, some are nocturnal and hide in crevices during the day (a tactic that is made far easier by the absence of a cumbersome shell), some can swim rapidly to escape danger, and certain species even have the remarkable ability to absorb intact stinging cells (nematocysts) from the cnidarians upon which they feed, and incorporate them into the slugs' own defences! In some sea slugs, secondary gills have evolved as outgrowths from the skin, since the mantle cavity, and the original gills (ctenidia) that it housed, were lost along with the shell.
The cephalopods
In a phylum dominated by small browsers and filter- feeders, the cephalopods stand out as a group that have become exquisitely adapted to an actively-swimming, predatory lifestyle. In both size and intelligence, they are unrivalled among invertebrates, and they show a remarkable divergence from the typical molluscan design. With such a highly-derived body plan, it is perhaps not surprising that the cephalopods also show a clear trend towards reduction, internalisation and loss of the characteristic molluscan shell.
Cephalopods are believed to be derived from a gastropod-like ancestor, and the most primitive types do have external shells. To provide the buoyancy necessary for a pelagic existence, these cephalopods developed a system of gas-filled chambers within the shell. Early cephalopods diverged into three main groups: the nautiloids and ammonoids, which retained an external shell (often elaborately coiled), and the squid-like belemnoids, in which the shell became straightened and internalised. Today, the ammonoids are extinct, and the nautiloids reduced to a single genus (the eponymous Nautilus). The belemnoids, however, diversified into several surviving lineages, giving rise to modern cuttlefish, squid and octopods - collectively known as 'coleoids'.
The internalisation of the shell provides coleoids with several advantages, most of which are related to the development of more efficient swimming. (The coleoids may originally have evolved in response to increasing competition from predatory fish, with which they are convergent in many ways.) Liberation from an external shell helps the coleoids to float horizontally in the water, and has allowed the development of fins for better locomotion, and a highly contractible mantle cavity that can squirt out water violently, moving the animal by jet propulsion. The uncovering of the body has also permitted the evolution of 'chromatophores', colour-changing cells in the skin, which are important in many coleoids for communication or camouflage.
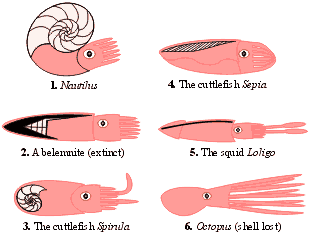
A simplified representation of the body plans of different cephalopod molluscs, illustrating the way in which the shell has been lost or reduced in many groups.
The extent of shell reduction varies between coleoids. The internal shell of a cuttlefish, although somewhat reduced in comparison to that of the early belemnoids, retains air spaces and continues to function in buoyancy control. In squid, by contrast, all that is left of the shell is a flat, cartilaginous 'pen', and in octopods it has disappeared completely. Squid are pelagic animals, and having lost their built-in float they are forced to maintain their buoyancy through biochemical tricks, such as retaining lightweight ammonia in their tissues in place of heavier ions. Their advantage over cuttlefish, however, is that they can colonise the deep oceans, where the pressure would cause a gas-filled shell to implode. Some deep-water squid grow to gigantic sizes, but their size alone cannot explain the trend towards reduction of the shell, since there are fossil examples of cephalopods with substantial shells that nonetheless managed to grow to monstrous proportions, such as the two-metre wide ammonite Pachydiscus and the straight-shelled, five-metre nautiloid Endoceras.
In many aspects of their anatomy and behaviour, the most advanced cephalopods are the octopods (Octopus and its relatives), and in this group the shell has been lost completely. Octopi have largely reverted to a benthic existence, where buoyancy is unnecessary, and the flexible, shell-less nature of their bodies allows them to squeeze into extremely tight crevices in pursuit of prey.
Conclusion
Shell loss and reduction has occurred in several separate groups of molluscs, occupying diverse niches in diverse habitats, so it is perhaps not surprising that the trait has no single, straightforward explanation. The many possible benefits of shell loss include weight reduction (in pelagic sea slugs), coping with mineral deficiency (in land slugs), streamlining (in swimming or burrowing species), an ability to crawl into tight spaces (in slugs and octopi), a more distensible body (in forms that eat large prey or swim by jet propulsion), and the exposure of the skin to the outside world (permitting the development of fins, colour-changing chromatophores, or even photosynthesis). Many shell-less molluscs have adapted to their nakedness by evolving novel features that carry out the functions previously performed by the shell (or by organs in the mantle cavity underneath). Thus, cephalopods have lightweight tissues for buoyancy, land slugs have water-conserving habits, many sea slugs have developed secondary gills for gas exchange, and different molluscs have come up with a range of means to defend themselves against predators, including poisons and camouflage.
Evolutionary biology teaches us that there is no one perfect body design: a feature that suits one lifestyle may not suit another. The global success of the molluscs must therefore be attributed both to their ability to grow a protective shell, and to their ability to lose that shell when circumstances demand.
References include
Adamo S A & R T Hanlon, 1996. Do cuttlefish (Cephalopoda) signal their intentions to conspecifics during agonistic encounters? Animal Behaviour 52(1): 73-81.
Barnes R S K, P Calow, D W Golding, P J W Olive & J I Spicer, 2001. The Invertebrates: a synthesis.
Barnes & Ruppert, 1994. Invertebrate Zoology.
Boardman R S, A H Cheetham & A J Rowell, 1987. Fossil Invertebrates.
Bonnaud L, R Boucher-Rodoni & M Monnerot, 1995. Phylogeny of Cephalopods Inferred from Mitochondrial DNA Sequences. Molecular Genetics and Evolution 7(1): 44-54.
Bouchet P & Á Valdéz, 1998. Naked in toxic fluids: A nudibranch mollusc from hydrothermal vents. Deep Sea Research Part II: Topical Studies in Oceanography 45(1-3): 319-327.
Clarke M R & E R Trueman, 1988. The Mollusca, Volume 12: Palaeontology and Neontology of Cephalopods.
Conway Morris, S, 1998. The Crucible of Creation.
Forsythe J W, R T Hanlon & D E Joneschild, 1999. Crypsis, conspicuousness, mimicry and polyphenism as antipredator defences of foraging octopuses on Indo-Pacific coral reefs, with a method of quantifying crypsis from video tapes. Biological Journal of the Linnean Society 66(1): 1-22.
Futuyma D J, 1998. Evolutionary Biology.
Hausdorf B, 2001. Macroevolution in progress: competition between semislugs and slugs resulting in ecological displacement and ecological release. Biological Journal of the Linnean Society 74(3): 387-395.)
Morton J E, 1979. Molluscs.
|
"If the Creator had indeed lavished his best design on the creature he shaped in his own image, creationists would surely have to conclude that God is really a squid." Jared Diamond |
This was originally written as a university biology essay