Parasite Damage and Host Responses
The example of Plasmodium
Introduction
A parasite is an organism that lives on or inside another organism and depends upon its host's resources. Sometimes this relationship is benign, but often parasites cause severe damage to their hosts. In this essay I will illustrate the varied and complex ways in which such damage can arise, and the role that the host's immune system plays in the process, by focusing on one particular parasite: the protozoan Plasmodium, which causes malaria. Collectively, our species suffers damage due to this parasite on a horrific scale: each year, around 100 million people develop clinical malaria, and between 1.5 and 2.7 million die of the disease (Hommel & Gilles 1998).
The life cycle of Plasmodium involves an alternation between mosquito and vertebrate hosts. A mosquito acquires the parasite when it feeds upon the blood of an infected animal. Plasmodium undergoes sexual reproduction in the gut of the mosquito; the offspring then migrate to the insect's salivary glands, and are transferred into a vertebrate host when the mosquito feeds. In vertebrates, the parasites undergo successive rounds of asexual multiplication, first inside the host's liver cells and later in red blood cells (Knell 1991).
In the mosquito, Plasmodium is fairly benign, but in its vertebrate hosts it causes severe illness, characterised by periodic bouts of intense fever. Malaria caused by P. falciparum, one of the four Plasmodium species that infect humans, can produce life-threatening complications such as cerebral malaria (in which the parasite affects the brain), renal failure and pulmonary oedema (Hommel & Gilles 1998). Malaria is also associated with conditions such as anaemia and hypoglycaemia, which can have a severe impact upon health and survival, particularly among children and pregnant women (Mendis & Carter 1995).
The symptoms of malaria are the result of complex interactions between Plasmodium and the immune system of its host (Clark & Cowden 2003). Although the biology of the parasite is now well understood (Knell 1991), the exact mechanisms by which it causes disease are the subject of extensive controversy and ongoing research. The main processes that are believed to be involved are outlined in this essay, and illustrated in Figure 1.
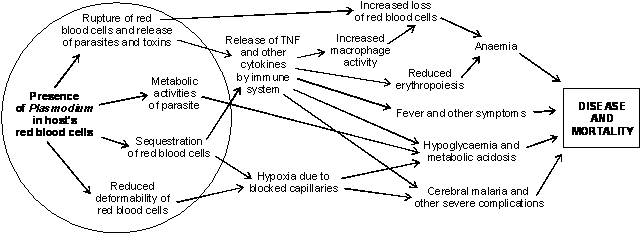
Figure 1: possible mechanisms by which Plasmodium causes disease
The immune response
The periodic bouts of fever that occur in malaria coincide with the synchronised rupture of Plasmodium-infected red blood cells. This causes the release of parasites en masse into the blood stream, along with pigments and toxins that have accumulated inside the red blood cells as a result of the parasites' metabolic activities. The presence of large quantities of parasite material in the blood triggers a dramatic immune response, mediated by the secretion of cytokine molecules by the cells of the immune system (Hommel & Gilles 1998). Some cytokines—such as 'tumour necrosis factor' (TNF), interferon-gamma, interleukin-12 and interleukin-18—enhance the immune response, stimulating macrophages and other immune cells to destroy parasites by phagocytosis and by the production of toxins. Other cytokines—including interleukin-4, interleukin-10 and TGF-beta—help to regulate the immune response by dampening these effects (Malaguarnera & Musumeci 2002).
Although the immune response stimulated by cytokines undoubtedly plays an important role in suppressing and killing malaria parasites within the body, excessive production of cytokines can have pathological consequences (Richards 1997). The high levels of TNF and other 'inflammatory' cytokines produced in response to Plasmodium are responsible for the intense fever associated with malaria. Plasmodium triggers the same immune pathways as a variety of other parasites, which may explain why the symptoms of malaria closely resemble those of many other diseases (Clark & Cowden 2003). It is not known whether malarial fever is an adaptive response by the host that helps to kill the parasites (Mendis & Carter 1995, Ramasamy 1998), or a pathological reaction caused by over-stimulation of the immune system (Hommel & Gilles 1998). Overproduction of inflammatory cytokines may also be responsible for the life-threatening complications such as cerebral malaria that occur in a small proportion of malaria patients (Mendis & Carter 1995).
Sequestration
Red blood cells infected with Plasmodium falciparum display protein-rich 'knobs' on their outer surfaces, which cause the cells to adhere to one another and to capillary walls. This adhesion allows parasite-infected cells to remain 'sequestered' in particular organs rather than circulating freely in the bloodstream, helping the parasite to evade the host's immune system. Since almost all of the human deaths attributed to malaria are caused by P. falciparum—the only human-infecting Plasmodium species with the ability to sequester—it is widely believed that that sequestration plays a key role in cerebral malaria and other fatal complications of the disease (Ramasamy 1998). However, there are conflicting theories about how this occurs.
The traditional explanation for cerebral malaria is that sequestration, perhaps combined with the reduction in the deformability of red blood cells that occurs when the cells are infected with Plasmodium, leads to the blockage of capillaries in the brain, depriving the tissue of oxygen (Dondorp et al 2000). However, measurements made using Near Infrared Spectroscopy and Doppler sonography show that levels of blood flow in the brains of cerebral malaria patients are not abnormally low (Mendis & Carter 1995), and individuals who recover from cerebral malaria do not generally exhibit the permanent brain damage that is typically associated with acute oxygen deprivation. It has therefore been suggested that sequestered cells infected with P. falciparum harm the brain by causing an excessive immune reaction there, rather than by physically blocking capillaries. Nitric oxide (NO), a substance that is manufactured by macrophages to kill parasites but is also toxic to host cells at high concentrations, has been implicated in this damaging immune reaction. It has been reported that a toxin produced by P. falciparum can induce the NO-synthesising enzyme iNOS in human endothelial cells, and iNOS has been found in samples of brain tissue taken during autopsies of cerebral malaria victims (Clark et al 1997).
In pregnant women, P. falciparum frequently sequesters in the placenta, where rich capillary beds and weakened immune responses create a hospitable environment for the parasite. Placental malaria can have harmful consequences for the foetus, disrupting its supply of oxygen and nutrients, and increasing the risk of premature delivery. (Menendez 1995, Nosten et al 2004). These problems cause babies born to malaria-infected mothers to have an unusually high probability of low birth weight, which in turn is associated with higher levels of infant mortality (Guyatt & Snow 2001).
Anaemia
Plasmodium, like many parasites, can damage its host by causing anaemia—a reduction in the ability of the blood to transport oxygen, which leads to lethargy and (in very extreme cases) can be fatal. The decrease in red blood cell concentration that is responsible for malarial anaemia occurs both through an increase in the rate at which red blood cells are destroyed and a decrease in the rate at which new ones are produced. Plasmodium not only causes the rupture of parasitized red blood cells, but stimulates the activity of macrophages in the spleen, which then destroy both parasitized and unparasitized red blood cells. (During malaria infection, unparasitized red blood cells may be targeted because they have abnormally rigid membranes, or because malarial antigens present in the bloodstream bind to their surfaces.) TNF-alpha and other cytokines produced during malaria depress the rate of erythropoiesis (the manufacture of new red blood cells), further contributing to anaemia (Menendez et al 2000). The health risks due to malaria-related anaemia are particularly severe in pregnant women, and there is some evidence that maternal anaemia is associated with anaemia in the foetus, which increases the risk of infant mortality (Menendez 1995).
Physiological changes
Parasites can produce physiological abnormalities in their host, which may have harmful consequences throughout the body, not just in the tissues or organs in which the parasite is present. Malaria patients commonly exhibit hypoglycaemia—a particular problem in pregnant women, in whom hypoglycaemia may be a cause of low foetal birth weight (Menendez 1995)—and metabolic acidosis, which is the cause of a significant number of malaria-related fatalities among young children (Mendis & Carter 1995). Possible explanations for these physiological changes include anaerobic consumption of glucose by the parasites, and hypoxia due to the blockage of capillaries by parasite-infected red blood cells. There is also strong evidence that the cytokines produced during malaria can cause hypoglycaemia and acidosis by inducing changes in the body's carbohydrate metabolism. Hypoglycaemia and acidosis can be induced in the absence of malaria by injecting TNF into animals, and in human malaria patients TNF levels correlate with hypoglycaemia (Clark et al 1997, Clark & Cowden 2003).
Factors affecting the severity of disease
A given variety of parasite will not affect all hosts in an identical way. The likelihood of parasite infection being established and the severity of the damage caused depend upon many factors, included the host's genotype, age, nutritional status and immunological history.
It has been known for a long time that individuals with certain genotypes, such as those carrying the famous 'sickle-cell' haemoglobin allele, are protected against infection by Plasmodium. More recently, researchers have also discovered genetic variations among humans that affect the severity of the damage caused when Plasmodium infection does become established (Mendis & Carter 1995). For example, a particular allele of the TNF-alpha gene-promoter region was found to be associated with a high probability of developing fatal cerebral malaria in patients infected with P. falciparum (Malaguarnera & Musumeci 2002).
The outcome of an infection also depends upon the genotype of the parasite. An obvious (albeit somewhat unnatural) illustration of this is provided by the existence of localised varieties of P. falciparum that show an inherited resistance to certain anti-malarial drugs (Knoll 1991, Mendis & Carter 1995).
People living in areas where malaria is highly endemic, who are repeatedly infected with Plasmodium, eventually attain a 'semi-immune' state in which an uneasy balance is reached between the parasite and the host's immune system. Such people continue to harbour significant levels of Plasmodium in their blood, but do not show obvious symptoms of disease. Children do not normally acquire this semi-immunity until around the age of five, which partly explains why young children account for the vast majority of deaths that occur due to malaria (Mendis & Carter 1995). Factors that weaken the immune system—such as pregnancy (Shulman & Dorman 2003), micronutrient malnutrition (Nussenblatt & Semba 2002), and HIV infection (Rowland-Jones & Lohman 2002)—have been associated with an increased incidence of Plasmodium infection, and with a resurgence of symptoms in those chronically infected with the parasite.
Conclusion
Parasites may harm their hosts by causing physical damage (such as the destruction of host cells or the blockage of blood vessels) or by triggering unpleasant physiological changes (such as the induction of fever). Some harmful effects result directly from parasites' activities, while others are side-effects of the mechanisms by which the host's immune system attempts to kill the parasites. In the case of malaria (and many other parasitic diseases), it is increasingly recognised that effects of the cytokines released by the host's immune system in response to the parasite are responsible for many of the symptoms of disease.
In order to limit the damage done by an invading parasite, a host's immune system must respond in a balanced and well-regulated manner. A response that is too weak will fail to rid the host of a parasite infection. However, as the example of malaria demonstrates, an excessive or inappropriate immune response may exacerbate the harm that a parasite causes to its host.
References
I.A. Clark, F.N. Al Yaman & L.S. Jacobson (1997), The Biological Basis of Malarial Disease. International Journal for Parasitology, vol. 27, no. 10, pp. 1237-1249.
I.A. Clark & W.B. Cowden (2003), The pathophysiology of falciparum malaria. Pharmacology & Therapeutics, vol. 99, pp. 221– 260.
A.M. Dondorp, P.A. Kager, J. Vreeken & N.J. White (2000), Abnormal Blood Flow and Red Blood Cell Deformability in Severe Malaria. Parasitology Today, vol. 16, no. 6, pp. 228-232.
H.L. Guyatt & R.W. Snow (2001), Malaria in pregnancy as an indirect cause of infant mortality in sub-Saharan Africa. Transactions of the Royal Society of Tropical Medicine and Hygiene, vol. 95, pp. 569-576.
M. Hommel & H. M. Gilles (1998), Malaria. In Topley & Wilson's Microbiology and Microbial Infections edited by L. Collier, A. Balows & M. Sussman, Ninth Edition, Vol. 5 Parasitology, edited by F.E.G. Cox, J.P. Kreier & D. Wakelin, chapter 20. Arnold.
A.J. Knell (1991), Malaria. Oxford University Press.
L. Malaguarnera & S. Musumeci (2002), The immune response to Plasmodium falciparum malaria. The Lancet Infectious Diseases, vol. 2, pp. 472-478.
K.N. Mendis & R. Carter (1995), Clinical Disease and Pathogenesis in Malaria. Parasitology Today, vol. 11, no. 5, pp. 2-16.
C. Menendez (1995), Malaria During Pregnancy: A Priority Area of Malaria Research and Control. Parasitology Today, vol. 11, no. 5, pp. 178-183.
C. Menendez, A.F. Fleming & P.L. Alonso (2000), Malaria-related Anaemia. Parasitology Today, vol. 16, no. 11, pp. 469-476.
F. Nosten, S. J. Rogerson, J.G. Beeson, R. McGready, T.K. Mutabingwa & B. Brabin (2004), Malaria in pregnancy and the endemicity spectrum: what can we learn? Trends in Parasitology, vol. 20, no. 9, pp. 425-432.
V. Nussenblatt & R.D. Semba (2002), Micronutrient malnutrition and the pathogenesis of malarial anemia. Acta Tropica, vol. 82, pp. 321–337.
R. Ramasamy (1998), Molecular basis for evasion of host immunity and pathogenesis in malaria. Biochimica et Biophysica Acta, vol. 1406, pp. 10–27.
A.L. Richards (1997), Tumour Necrosis Factor and Associated Cytokines in the Host’s Response to Malaria. International Journal for Parasitology, vol. 27, no. 10. pp. 1251-1263.
S.L. Rowland-Jones & B. Lohman (2002), Interactions between malaria and HIV infection—an emerging public health problem? Microbes and Infection, vol. 4, pp. 1265–1270.
C.E. Shulman & E.K. Dorman (2003), Reducing childhood mortality in poor countries: Importance and prevention of malaria in pregnancy. Transactions of the Royal Society of Tropical Medicine and Hygiene, vol. 97, pp. 30-35.
This was originally written as a university biology essay